
The pharmaceutical and biotechnology industries are increasingly reliant on robust data collection and analysis to ensure the safety and efficacy of clinical trials. A critical component of this process is the creation and maintenance of comprehensive monitoring reports. These reports provide a detailed account of the clinical trial's progress, highlighting key metrics, potential issues, and overall performance. A well-structured monitoring report template is essential for informed decision-making, regulatory compliance, and stakeholder communication. This article will delve into the key elements of a robust monitoring report template, offering practical guidance for clinical trial teams and stakeholders alike. Monitoring Report Template Clinical Trials is more than just a document; it's a vital tool for optimizing trial design, identifying and mitigating risks, and ultimately, delivering valuable insights to patients. The goal is to provide a clear, concise, and actionable record of the trial's journey, enabling proactive adjustments and ensuring the integrity of the data.
Understanding the Importance of Monitoring Reports
The initial stages of a clinical trial are often characterized by uncertainty. Data collection can be challenging, and unforeseen events can occur. A comprehensive monitoring report provides a framework for systematically tracking progress, identifying deviations from the planned trajectory, and taking corrective action. Without a standardized reporting system, it becomes difficult to assess the trial's performance objectively and make informed decisions about future studies. Furthermore, regulatory agencies require detailed reports to ensure patient safety and data integrity. Failure to meet these requirements can lead to delays, penalties, and reputational damage. Therefore, investing in a robust monitoring report template is a strategic investment in the success of any clinical trial.

Core Components of a Monitoring Report Template
A comprehensive monitoring report typically includes several key sections. Each section is designed to address a specific aspect of the trial's progress. Let's examine some of the most important components:
1. Trial Overview and Objectives
This initial section provides a concise summary of the trial's purpose, design, and key objectives. It should clearly state the intended population, intervention, endpoints, and primary and secondary outcomes. It's crucial to reiterate the trial's goals and the rationale behind them. For example, "This study aims to evaluate the efficacy of Drug X in treating Disease Y in patients with condition Z, as determined by the primary endpoint of…" This section serves as a foundational document for all subsequent reports. Monitoring Report Template Clinical Trials emphasizes the importance of accurately reflecting the trial's original design and objectives.
2. Participant Data and Safety
This section is arguably the most critical, focusing on the safety and well-being of the trial participants. It details the number of participants enrolled, demographics, baseline characteristics, adverse events (AEs), and serious adverse events (SAEs). Detailed information on participant adherence to the protocol, including any challenges encountered, is also crucial. Regular monitoring of safety data is essential to identify potential risks and implement appropriate mitigation strategies. The template should include a clear process for reporting and investigating AEs and SAEs, adhering to established guidelines. Monitoring Report Template Clinical Trials highlights the need for meticulous documentation of participant data and a proactive approach to safety monitoring.
3. Clinical Observations and Assessments
This section focuses on the ongoing assessment of participants' clinical status. It includes observations related to symptoms, functional status, and overall well-being. This may involve regular assessments by clinical staff, questionnaires, and potentially, physical examinations. The frequency and type of assessments should be clearly defined and documented. Furthermore, the template should address any changes in participant symptoms or functional status that warrant further investigation. Monitoring Report Template Clinical Trials stresses the importance of consistent and objective observation of participants.
4. Protocol Deviations and Changes
This section meticulously documents any deviations from the planned protocol, including changes in study procedures, participant enrollment, or data collection methods. It's vital to record the rationale for any deviations and the impact on the trial's progress. A clear process for managing protocol changes is essential to maintain the integrity of the data and ensure the trial remains on track. Monitoring Report Template Clinical Trials underscores the need for a robust system for tracking and documenting protocol deviations.

5. Data Management and Quality Control
This section addresses the procedures for data collection, management, and quality control. It covers aspects such as data entry, validation, and quality assurance measures. The template should specify the data management system used, data quality control procedures, and the roles and responsibilities of data managers. Ensuring data integrity is paramount to the reliability of the monitoring report. Monitoring Report Template Clinical Trials emphasizes the importance of a strong data management infrastructure.
6. Financial and Regulatory Reporting
This section provides a summary of all relevant financial and regulatory reporting requirements. It includes details on payments to participants, expenses related to the trial, and any regulatory submissions. It also addresses compliance with applicable regulations, such as Good Clinical Practice (GCP) and data privacy regulations. Maintaining accurate and complete financial and regulatory reporting is crucial for ensuring the trial's compliance and avoiding penalties.
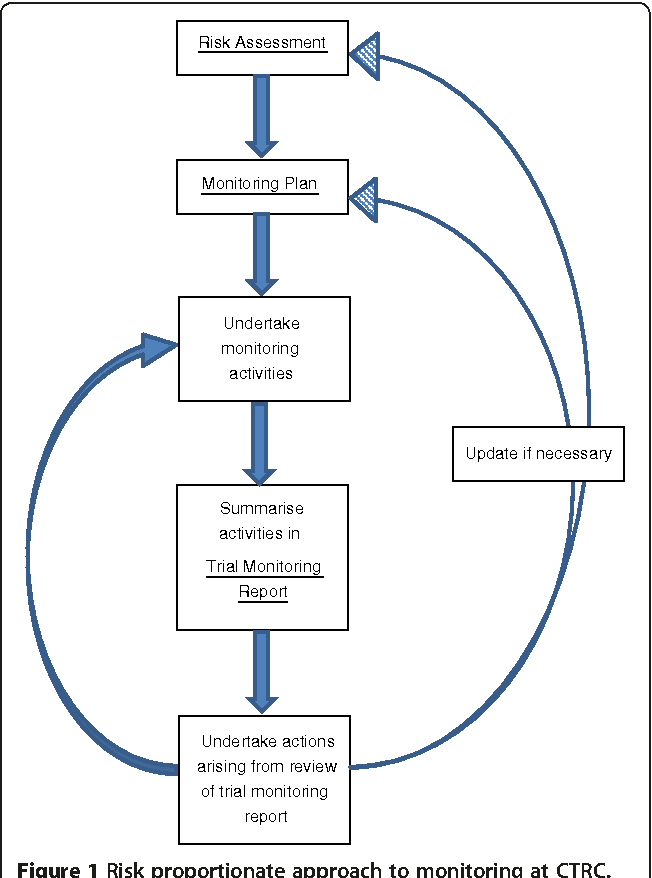
The Role of Statistical Analysis in Monitoring Reports
Beyond simply recording data, a strong monitoring report incorporates statistical analysis to identify trends and patterns. This may involve calculating descriptive statistics, performing trend analysis, and using statistical tests to assess the significance of observed changes. The template should include sections for reporting on key statistical metrics, such as the incidence of adverse events, the proportion of participants meeting specific criteria, and the overall efficacy of the intervention. Monitoring Report Template Clinical Trials highlights the increasing importance of statistical analysis for interpreting trial data.
Best Practices for Effective Monitoring Reports
Creating an effective monitoring report requires adherence to best practices. Here are some key considerations:

- Timeliness: Reports should be generated and distributed promptly, ideally within 24-48 hours of significant events.
- Clarity and Conciseness: Use clear and concise language, avoiding jargon and technical terms that may not be understood by all readers.
- Visualizations: Incorporate charts and graphs to effectively communicate data trends and insights.
- Accuracy: Ensure all data and information presented in the report are accurate and verifiable.
- Accessibility: Make the report easily accessible to all relevant stakeholders.
Conclusion
Monitoring reports are an indispensable tool for the success of any clinical trial. They provide a comprehensive and objective record of the trial's progress, enabling informed decision-making, regulatory compliance, and patient safety. By implementing a well-structured monitoring report template and adhering to best practices, clinical trial teams can significantly improve the likelihood of delivering valuable insights and ultimately, improving patient outcomes. The continued evolution of data analytics and statistical methods will further enhance the value of monitoring reports, providing even more sophisticated tools for optimizing clinical trial design and execution. Monitoring Report Template Clinical Trials is a dynamic document, constantly adapting to the evolving needs of the pharmaceutical and biotechnology industries.

0 Response to "Monitoring Report Template Clinical Trials"
Posting Komentar